Production method of monoethanolamine

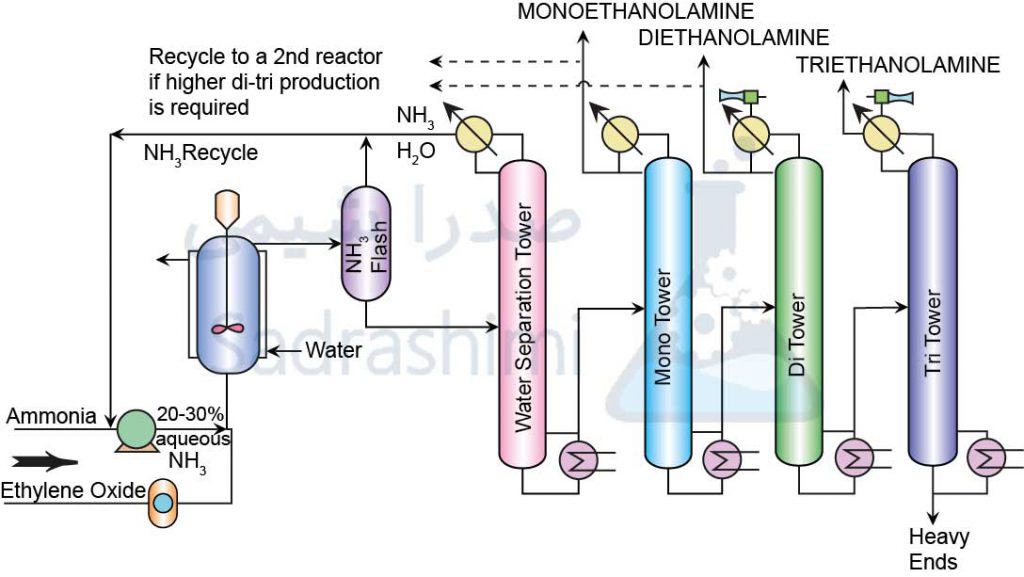
Monoethanolamine is obtained from the reaction between ammonia and ethylene oxide. This reaction produces diethanolamine and triethanolamine in addition to monoethanolamine. The production ratio of different products can be adjusted by changing the stoichiometry of the reaction. This reaction is thermal and should prevent the temperature from rising and reaching the flash point. Ethylene oxide reacts slowly with ammonia, adding water to speed up the reaction. In processes without water, a fixed bed catalytic reactor with ion exchange resins or zeolite and clay is used. The whole process takes place in the liquid phase and the pressure in the reactor is high enough to prevent ethylene oxide and ammonia from evaporating at the reaction temperature.
Monoethanolamine is recommended as a substance for making alkanolamides because of its reaction with fatty acid or coconut oil. As a result of its properties, this product is used in various industrial sectors such as detergents, lubricating oils, health and personal care products, mineral suspensions, etc. printer ink used in to control pH in the preparation of inks and dyeing and textile industries. Complementary textile processes complementary textile processes are generally referred to as the processes that take place after dyeing on fibers. Wood Polishing: MEA is used in both Quaternary Alkaline Copper (ACQ) and Azole Copper formulations, which are used in wood polishing to increase the wood's resistance to pests and erosion. Both compounds are arsenic-free alternatives to the terminated CCA formulation.

In the 1930s, alkanolamines were first used to sweeten gas, and from then until the 1970s, monoethanolamine (MEA) was most widely used, but since the 1970s, due to its disadvantages, such as monoethanolamine, such as corrosion and Solvent Loss, Diethanolamine (DEA) replaced this amine in the mid-1970s, and especially in the last two decades due to advantages such as the selective separation of hydrogen sulfide in the presence of carbon dioxide, high stability and Low energy consumption for solvent recycling has been widely used in the gas industry. Alkanolamines generally consist of at least one hydroxyl group (-COH) and at least one amino group (-NH2). Common amines used for gas sweetening are:
Primary, monoethanolamine (MEA) and diglycolamine (DGA) amines
Secondary Amines, Diethanolamine (DEA) and Diisopropanolamine (DIPA)
Tertiary, triethanolamine (TEA) and methyl diethanolamine (MDEA) amines
Methyl diethanolamine is a tertiary amine and therefore lacks active amino groups (-NH) in its molecular structure. Therefore, it has a stable chemical structure and selectivity for adsorption. In fact, the main factor in the decomposition of the first and second types of amines is the presence of these active amino groups. Decomposed amines are usually highly corrosive. This stability of the third type of amine allows its high concentrations (up to 3%) to be used without regard to corrosion problems. This case causes in order to absorb a certain amount of acid gases, the required flow rate of the third type of amine solution is less than the required flow rate of the first or second type of amine solution, and as a result, the amount of thermal energy consumed by regenerative tower reboilers is significantly reduced.
